Pune, India | November 25, 2025
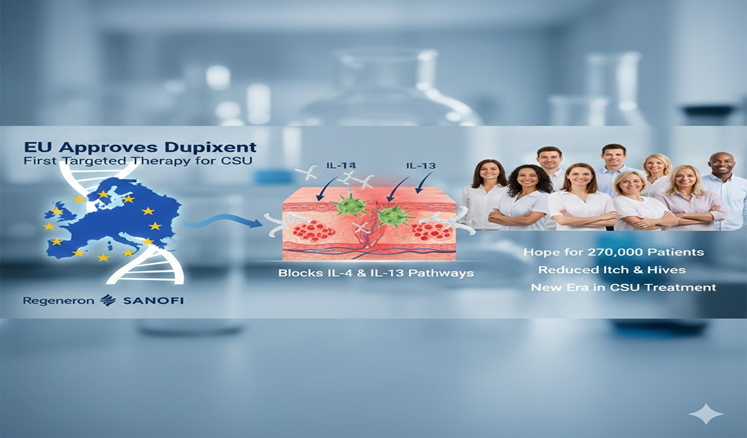
Regeneron Pharmaceuticals and Sanofi announced that the European Commission has officially approved Dupixent (dupilumab) as the first targeted therapy for moderate-to-severe chronic spontaneous urticaria (CSU) in individuals aged twelve years and older. This decision marks the first new targeted treatment for CSU in over a decade. It promises fresh hope to nearly 270,000 adolescents and adults across the European Union. These patients continue to experience persistent hives and itching even after undergoing standard antihistamine therapy.
The European Commission’s decision came after solid Phase 3 clinical trial data demonstrated Dupixent’s ability to significantly reduce both itch severity and hive count by the twenty-fourth week. When compared with a placebo group, Dupixent showed consistent improvements across several clinical measures. The treatment acts by blocking two key inflammatory pathways, interleukin 4 (IL-4) and interleukin 13 (IL-13). Thereby addressing the immune mechanisms responsible for driving chronic spontaneous urticaria.
George D. Yancopoulos, M.D., Ph.D., President and Chief Scientific Officer at Regeneron, described the approval as a milestone for individuals living with unpredictable and severe hives. He emphasized that Dupixent’s innovative mechanism provides physicians with a new first-line, targeted treatment alternative for CSU. According to him, such breakthroughs redefine treatment possibilities for patients who previously had few reliable medical options.
Furthermore, Tonya Winders, President and CEO of the Global Allergy & Airways Patient Platform, highlighted that many CSU sufferers struggle with sudden flare-ups, itching, and distressing skin reactions. She praised the European Commission’s approval, explaining that Dupixent offers hope for those who often endure limited symptom control from conventional therapies. In her statement, she expressed optimism that this drug could bring measurable improvements in patient comfort and daily functioning.
With this authorization, Dupixent now expands its portfolio to cover a seventh chronic inflammatory condition within the European Union. Previously, the medication had been approved for other type 2 inflammatory disorders. It includes atopic dermatitis in children aged six to eleven years. Such expansion reinforces Dupixent’s place as a leading biologic across multiple immune-related diseases, where excessive type 2 inflammation plays a central pathogenic role.
Industry analysts suggest that this new indication could solidify Dupixent’s dominance in the immunology sector. Its established safety profile and consistent efficacy across age groups contribute to physician confidence. Past pediatric studies have shown that common side effects included upper respiratory tract infections, injection site reactions, and conjunctivitis. Though overall tolerability remained stable across trials. These findings underline the drug’s suitability for continued long-term therapy under medical supervision.
According to its approved European label, Dupixent is administered through subcutaneous injection and can be self-injected following proper training. Regeneron and Sanofi confirmed that patients meeting eligibility criteria can now receive Dupixent as a first-line targeted treatment. This is particularly applicable to individuals with poor responses to standard antihistamines who have not yet started anti-IgE therapies. Consequently, the drug may offer a much-needed treatment bridge for an underserved patient population.
Regeneron reported that Dupixent was developed using its patented VelocImmune technology, which enabled the creation of fully human monoclonal antibodies that effectively block signaling through IL-4 and IL-13. This approach allowed researchers to precisely design biologics capable of modulating immune pathways implicated in allergic and inflammatory diseases. Moreover, the company views this approval as validation of Dupixent’s adaptability across various chronic, inflammation-driven medical conditions.
CSU remains a widespread condition throughout Europe. It is affecting a large number of patients who have faced years of frustration due to limited effective therapies. Because disease severity can fluctuate, access to a biological treatment that directly targets immune dysfunction represents a major paradigm shift. Physicians now possess an advanced therapeutic option to manage the root inflammatory causes rather than merely alleviating symptoms temporarily.
With this green light, healthcare professionals across European countries will gain access to a proven new tool in managing chronic spontaneous urticaria. Patients who previously depended solely on antihistamines can now experience a therapy reaching deeper into the molecular biology of their disease. Additionally, both Regeneron and Sanofi stated their plans to pursue expanded research investigating Dupixent’s potential. In other diseases where type 2 inflammation serves as a central trigger of symptoms.
Ultimately, the European Commission’s approval of Dupixent for chronic spontaneous urticaria signifies a major advancement for patient care and for medical science. This development not only fulfills critical unmet medical needs but also strengthens Dupixent’s standing as a pioneering biologic therapy in the broader fight against chronic inflammatory diseases. As the scientific community continues to unlock the mechanisms of immune dysregulation. Treatments like Dupixent highlight how targeted biologics can transform patient outcomes and redefine therapeutic standards across Europe and beyond.




