Pune, India | August 29, 2025
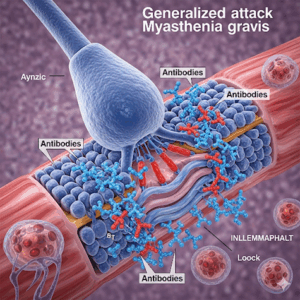
Regeneron Pharmaceuticals has recently announced promising Phase 3 trial results for cemdisiran. This RNA interference (RNAi) therapy specifically targets generalized myasthenia gravis (gMG), a rare autoimmune disorder that weakens skeletal muscles and affects daily activities. Importantly, the trial showed that cemdisiran met its primary endpoint by significantly improving key daily functions such as speaking, chewing, and swallowing over 24 weeks.
Furthermore, the study evaluated cemdisiran both as a standalone treatment and in combination with pozelimab, an FDA-approved C5 monoclonal antibody marketed as Veopoz. Patients receiving cemdisiran alone experienced a 74% reduction in C5 protein levels. Meanwhile, those on combination therapy saw a near 99% reduction. Notably, cemdisiran alone slightly outperformed the combination in improving daily function scores. This indicates that the RNAi treatment is effective even without additional antibody therapy.
Regarding safety, results were favorable. No patients discontinued treatment due to adverse effects. Based on these encouraging outcomes, Regeneron plans to file a Biologics License Application (BLA) with the FDA in early 2026. Consequently, this could potentially provide a new treatment option for people living with gMG.
Generalized myasthenia gravis disrupts communication between nerves and muscles. As a result, it causes muscle weakness in the face, limbs, eyes, and respiratory system. Cemdisiran targets the C5 protein, which is a critical element in the immune pathway responsible for this damage. Unlike many existing therapies that require frequent hospital visits, cemdisiran offers the convenience of subcutaneous dosing once every three months. Importantly, this can be done at home, which may significantly reduce the treatment burden for patients.
Regeneron views this simplified dosing schedule as a competitive advantage. In contrast, current treatments like Argenx’s Vyvgart Hytrulo, Johnson & Johnson’s Imaavy, and AstraZeneca’s Soliris require more frequent or clinic-based administration. Therefore, analysts suggest cemdisiran could capture a significant portion of the multibillion-dollar gMG treatment market. Its convenience and lasting effects make it especially appealing.
Following the trial announcement, Regeneron’s stock saw a modest increase. This reflects investor confidence in the company’s rare disease pipeline. Besides gMG, Regeneron is also investigating cemdisiran and pozelimab for other complement system-related conditions. These include paroxysmal nocturnal hemoglobinuria (PNH) and geographic atrophy, an eye disorder causing vision loss. Thus, this expands cemdisiran’s potential impact.
Cemdisiran is central to Regeneron’s strategy of targeting the complement system with advanced precision therapies. The RNAi mechanism silences the gene producing the C5 protein at the liver stage. Consequently, this offers sustained suppression without the need for continuous antibody treatment. While pozelimab uses a different mechanism, it complements cemdisiran’s effects. Together, they provide physicians with flexible treatment options.
Regeneron’s exploration of both monotherapy and combination regimens underscores its commitment to personalized care. The goal is quarterly dosing that improves patient outcomes and reduces emotional and practical challenges associated with treatment.
Beyond gMG, cemdisiran exemplifies Regeneron’s dedication to RNAi and gene-silencing technologies. This goes beyond conventional monoclonal antibodies. As a result, the innovation positions cemdisiran as a novel, patient-friendly therapy that could transform clinical management of gMG.
The trial results were well-received by healthcare providers and patient groups. They see cemdisiran as a promising option to improve the quality of life. Additionally, the therapy has a strong safety profile and ease of use. As Regeneron prepares for FDA submission, it is also gearing up for regulatory discussions worldwide. Meanwhile, the company is making supply chain arrangements and launching educational initiatives to support clinicians and patients.
With robust clinical data, a growing portfolio of targeted diseases, and continued investment in cutting-edge technologies, Regeneron is on track to launch the first RNAi therapy for gMG. Ultimately, the success of this Phase 3 trial strengthens Regeneron’s leadership in rare disease innovation. It offers new hope to thousands of patients who currently face limited treatment choices. By combining scientific excellence with a patient-centered approach, Regeneron advances therapies that aim to make a meaningful difference in patients’ lives.




